In a treat-through trial of adults with moderately to severely active CD1
Patients achieved BOTH early clinical response at week 12 AND key outcomes at Week 521,3
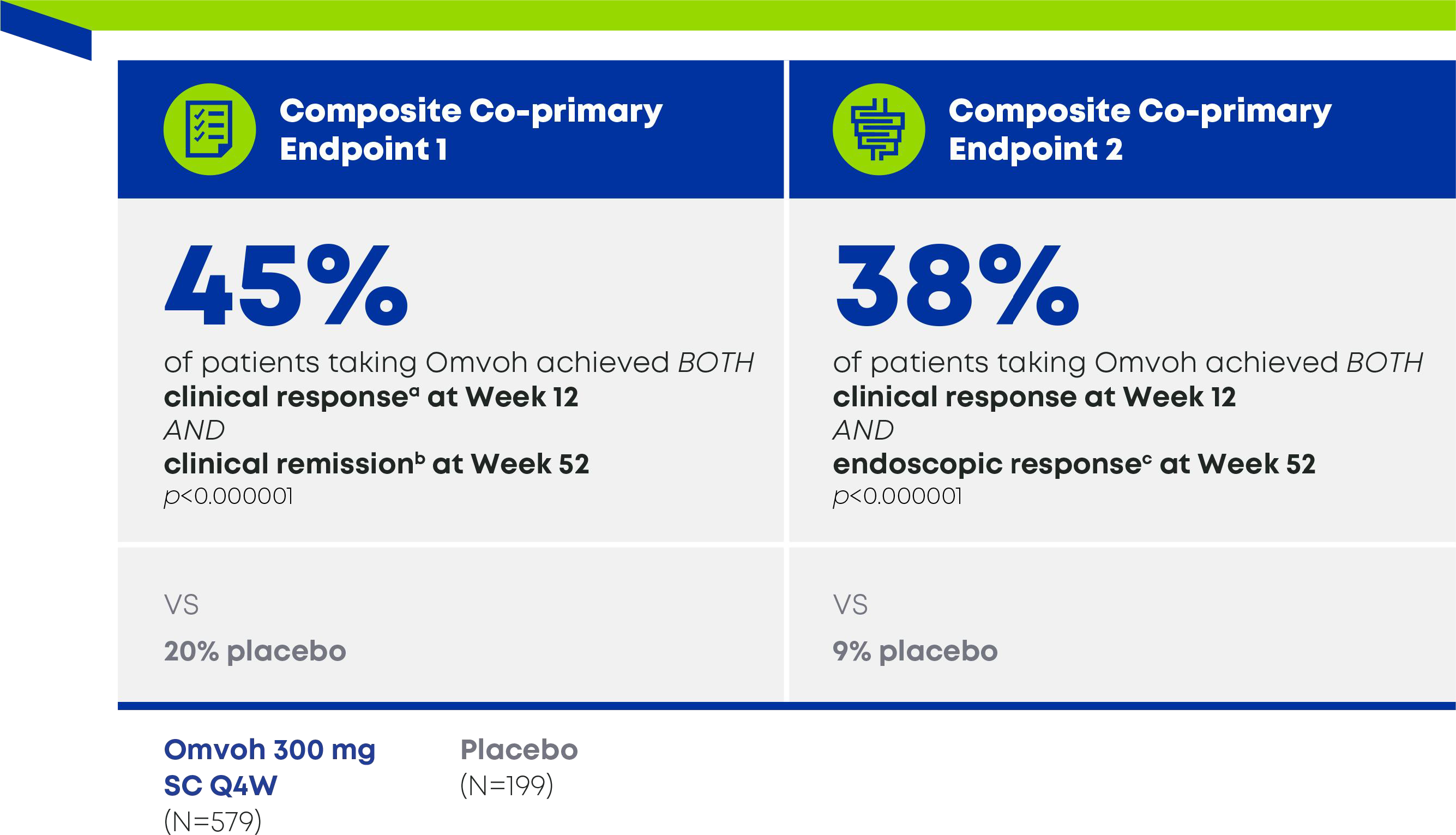
- a Clinical response by PRO was defined as ≥30% decrease in stool frequency and/or abdominal pain, and neither score worse than baseline.1
- bClinical remission was defined as a CDAI score of <150.1
- c Endoscopic response was defined as ≥50% reduction from baseline in SES-CD total score, based on central reading.1
- d Following mirikizumab 900 mg as an IV infusion at week 0, week 4, and week 8 patients received mirikizumab 300 mg as an SC injection at week 12 and every 4 weeks thereafter for up to an additional 40 weeks.1
CD=Crohn's Disease; CDAI=Crohn's Disease Activity Index; IV=intravenous; PRO=Patient-Reported Outcome; Q4W=every 4 weeks; SC=subcutaneous; SES-CD=Simple Endoscopic Score for Crohn's disease.
For adults with moderately to severely active CD1
Omvoh demonstrated early clinical response at week 12 and strong efficacy on meaningful outcomes at Week 521,3
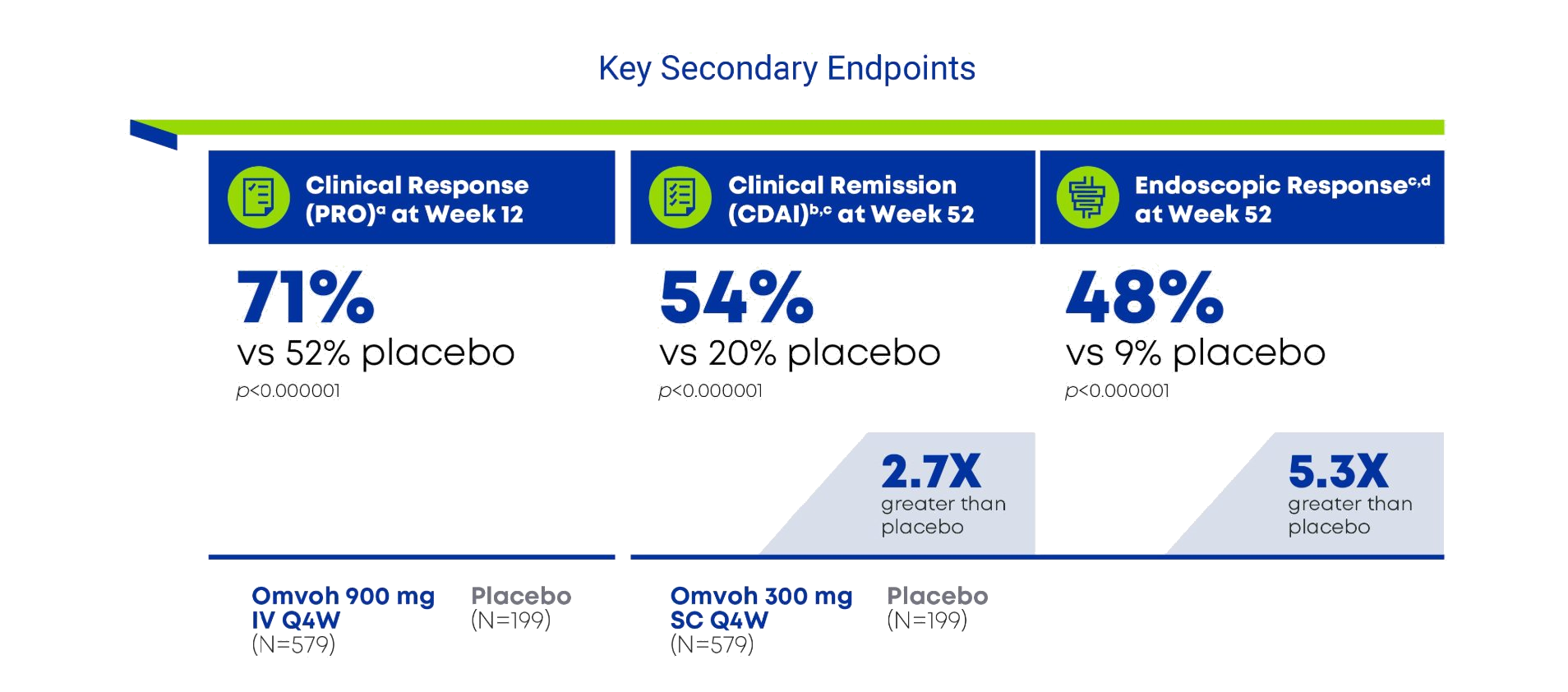
- a Clinical response by PRO was defined as ≥30% decrease in stool frequency and/or abdominal pain, and neither score worse than baseline.1
- bClinical remission was defined as a CDAI score of <150.1
- c Following mirikizumab 900 mg as an IV infusion at week 0, week 4, and week 8 patients received mirikizumab 300 mg as a SC injection at week 12 and every 4 weeks thereafter for up to an additional 40 weeks. Placebo sample size includes all patients randomized to placebo at baseline. Placebo patients that did not achieve clinical response by PRO at week 12 were considered non-responders at week 52.1
- d Endoscopic response was defined as ≥50% reduction from baseline in SES-CD total score. Placebo sample size includes all patients randomized to placebo at baseline. Patients that did not achieve clinical response by PRO at week 12 were considered non-responders at week 52.1
CDAI=Crohn's Disease Activity Index; IV=Intravenous; PRO=Patient-Reported Outcome; Q4W=Every 4 weeks; SC=Subcutaneous; SES-CD=Simple Endoscopic Score for Crohn's Disease.
For adults with moderately to severely active CD1
Demonstrated strong efficacy regardless of biologic experience3
Clinical Remissiona in Bio-naïveb and Bio-failedc Patients at Week 521,3
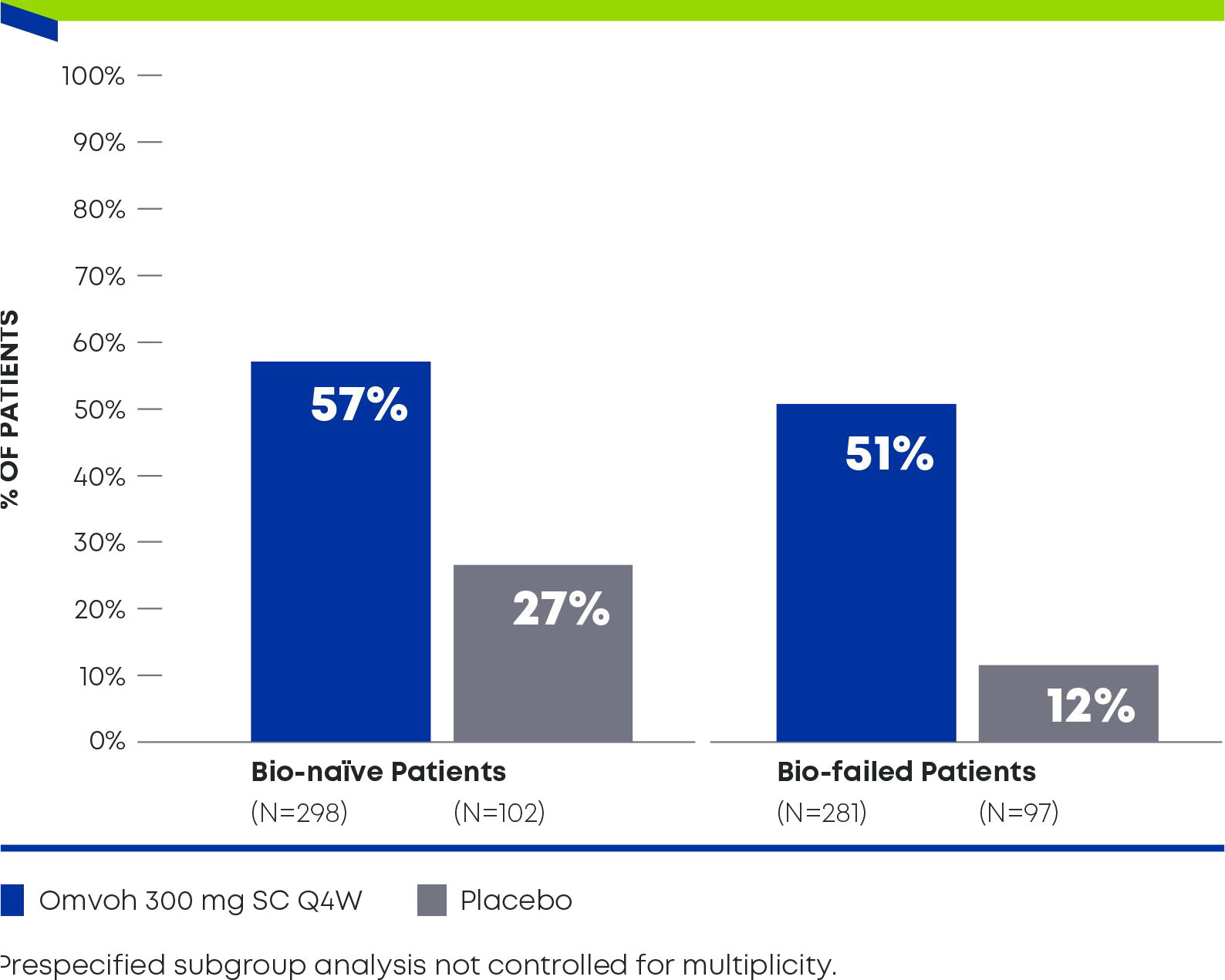
Omvoh in bio-naïveb:
57% achieved clinical remission and52% achieved endoscopic response3
Omvoh in bio-failedc:
51% achieved clinical remission and45% achieved endoscopic response3
Prespecified subgroup analysis not controlled for multiplicity.
- * Bio-naïve includes patients who either had not been treated with biologics or were exposed to biologics but did not experience inadequate response, loss of response, or intolerance.2
- † Prior biologic failure includes loss of response, inadequate response, or intolerance to one or more biologic therapy (e.g. TNF blocker or integrin receptor antagonist).2
- aClinical remission was defined as a CDAI score of <150.1
- b Bio-naïve includes patients who either had not been treated with biologics or were exposed to biologics but did not experience inadequate response, loss of response, or intolerance.1
- c Prior biologic failure includes loss of response, inadequate response, or intolerance to one or more biologic therapy (e.g. TNF blocker or integrin receptor antagonist).1
- d Following mirikizumab 900 mg as an IV infusion at week 0, week 4, and week 8 patients received mirikizumab 300 mg as a SC injection at week 12 and every 4 weeks thereafter for up to an additional 40 weeks.1
Bio-failed=biologic-failed; bio-naïve=biologic-naïve; CD=Crohn's disease; PRO=Patient-Reported Outcome; TNF=tumor necrosis factor.
For adults with moderately to severely active CD1
Demonstrated strong efficacy regardless of biologic experience3
Endoscopic Responsea in Bio-naïveb and Bio-failedc Patients at Week 521,3
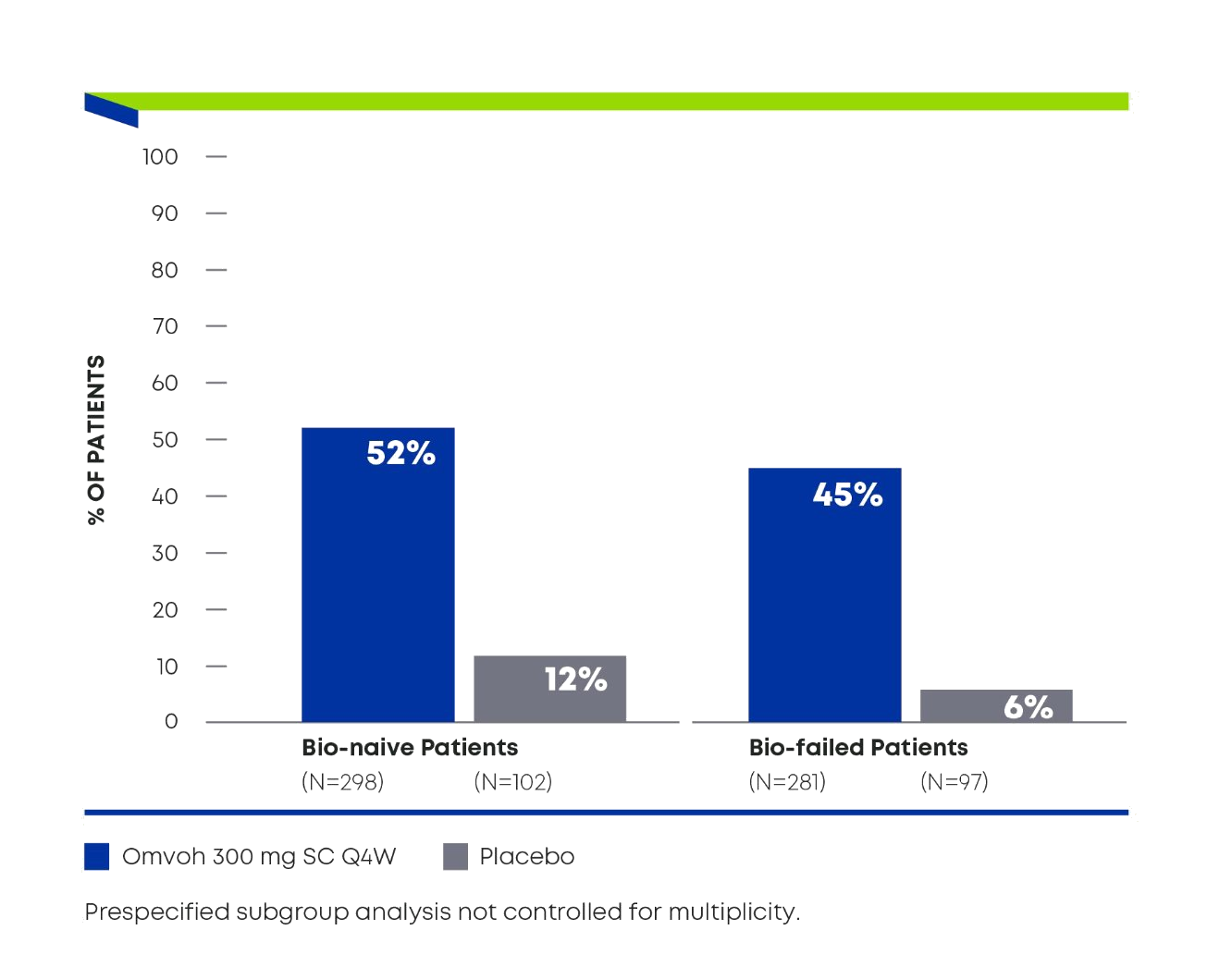
Omvoh in bio-naïveb:
57% achieved clinical remission and52% achieved endoscopic response3
Omvoh in bio-failedc:
51% achieved clinical remission and45% achieved endoscopic response3
Prespecified subgroup analysis not controlled for multiplicity.
- * Bio-naïve includes patients who either had not been treated with biologics or were exposed to biologics but did not experience inadequate response, loss of response, or intolerance.2
- † Prior biologic failure includes loss of response, inadequate response, or intolerance to one or more biologic therapy (e.g. TNF blocker or integrin receptor antagonist).2
- a Endoscopic response is defined as ≥50% reduction from baseline in SES-CD total score.1
- b Bio-naïve includes patients who either had not been treated with biologics or were exposed to biologics but did not experience inadequate response, loss of response, or intolerance.1
- c Prior biologic failure includes loss of response, inadequate response, or intolerance to one or more biologic therapy (e.g. TNF blocker or integrin receptor antagonist).1
- d Following mirikizumab 900 mg as an IV infusion at week 0, week 4, and week 8 patients received mirikizumab 300 mg as a SC injection at week 12 and every 4 weeks thereafter for up to an additional 40 weeks.1
Bio-failed=biologic-failed; bio-naïve=biologic-naïve; CD=Crohn's disease; PRO=Patient-Reported Outcome; TNF=tumor necrosis factor.
For adults with moderately to severely active CD1
About 90% of patients taking Omvoh maintained lasting efficacy through 2 years (Week 104)3
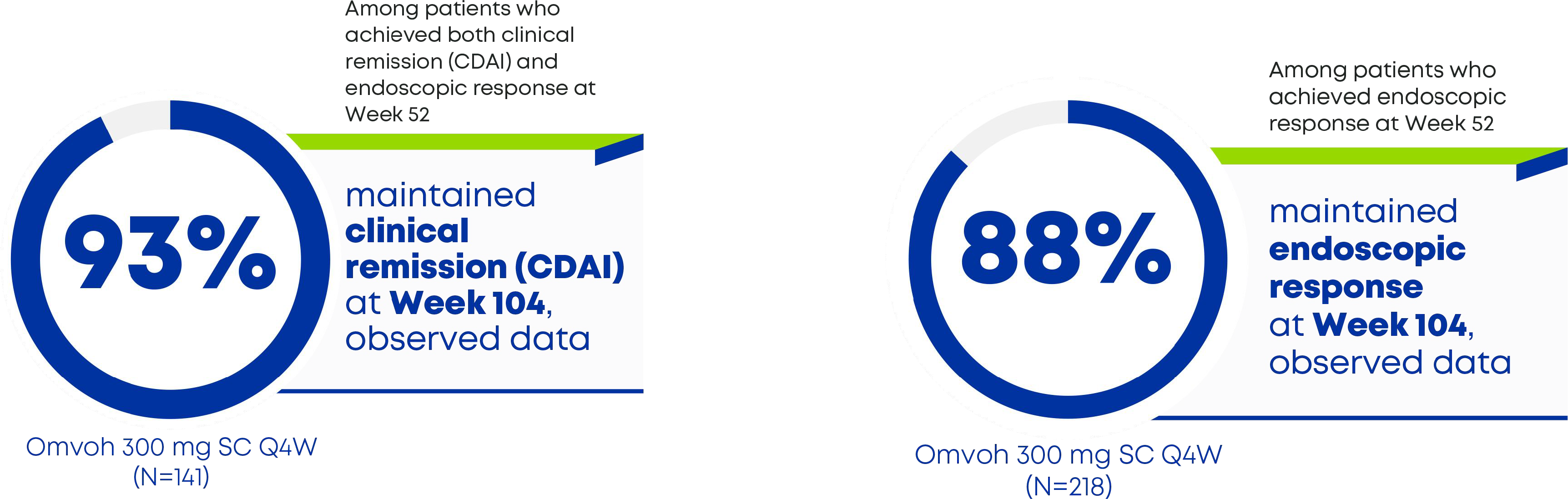
- Clinical remission was defined as a CDAI score of <150.
- Endoscopic response was defined as ≥50% reduction from baseline in SES-CD total score, based on central reading.
CD=Crohn's disease; CDAI=Crohn's Disease Activity Index; SES-CD=Simple Endoscopic Score for Crohn's Disease.
CD = Crohn's disease; CDAI = Crohn's Disease Activity Index; IV = intravenous; PRO = Patient-Reported Outcome; Q4W = every 4 weeks; SC = subcutaneous; SES-CD = Simple Endoscopic Score for Crohn's Disease; TNF = tumor necrosis factor.
- Omvoh EUSPC FEB 2025.
- Ferrante M, D'Haens G, Jairath V, Danese S, Chen M, Ghosh S, et al. Efficacy and safety of mirikizumab in patients with moderately-to-severely active Crohn's disease: a phase 3, multicentre, randomised, double-blind, placebo-controlled and active-controlled, treat-through study. Lancet. 2024;404(10470):2423–2436. doi.org/10.1016/S0140-6736(24)01762-8.
- Barnes EL, Sands BE, D'Haens G, Hisamatsu T, Kelly CR, Lu N, et al. Long-term efficacy and safety of mirikizumab following 104 weeks of continuous treatment for Crohn's disease: results from the VIVID-2 open-label extension study. CCC; February 6–8, 2025; San Francisco, CA.
