For adults with moderately to severely active UC1
Nearly 2 in 3 patients taking Omvoh achieved clinical response at Week 121
63% of patients taking Omvoh achieved clinical response after 12 weeks of induction dosing, and nearly 1 in 4 achieved clinical remission.
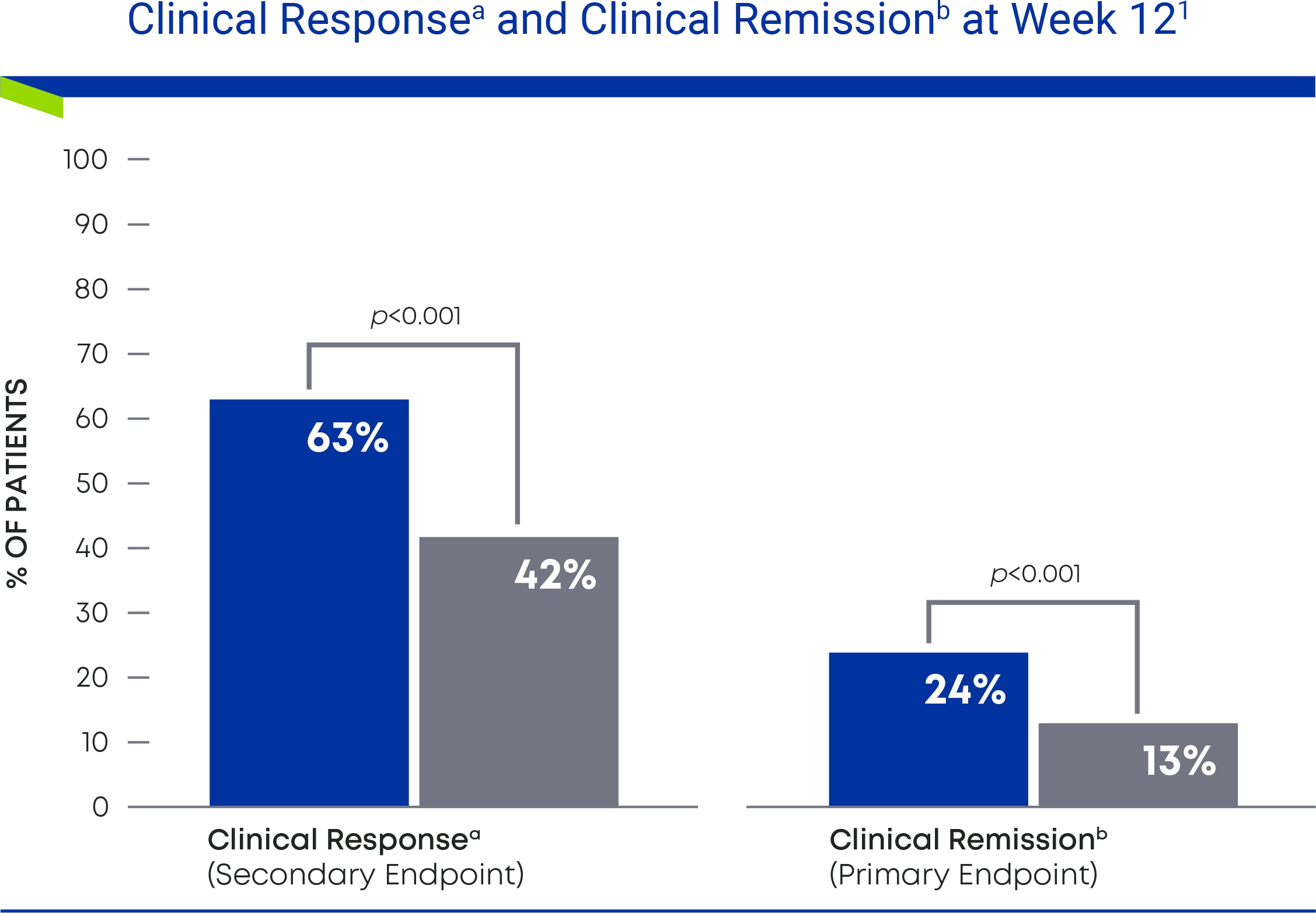
- a Clinical response was defined as a decrease in the modified Mayo Score (MMS) of ≥2 points and ≥30% from baseline, with either a decrease in rectal bleeding subscore of ≥1 or an absolute rectal bleeding subscore of 0 or 1.
- b Clinical remission was defined as an MMS subscore of stool frequency = 0 or 1 (with ≥1-point decrease from baseline), rectal bleeding = 0, and endoscopic = 0 or 1 (excluding friability).
IV=intravenous; MMS=modified Mayo Score; Q4W=every 4 weeks; UC= ulcerative colitis.
Among patients who achieved clinical response with Omvoh at Week 121
Omvoh demonstrated sustained clinical remission and mucosal healing at Week 521
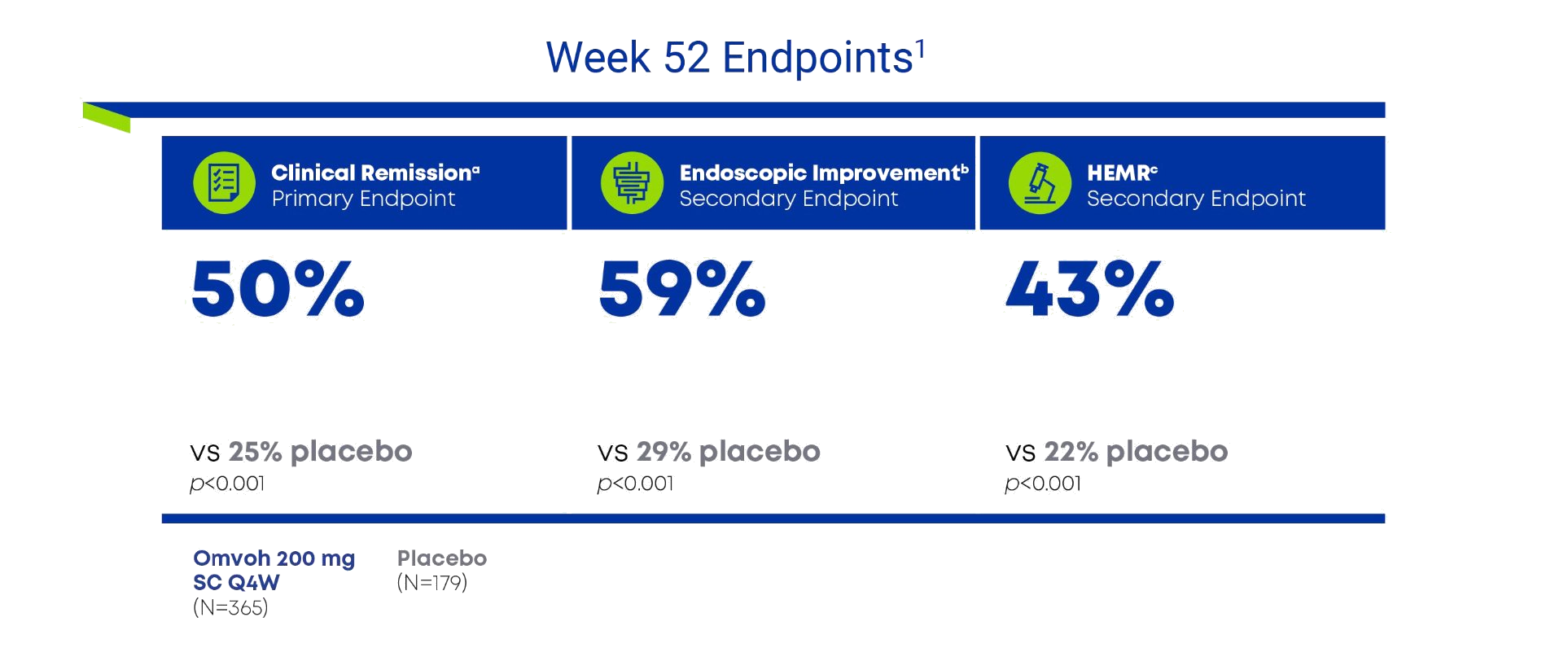
Post Hoc Analysis2
98% of patients who achieved clinical remission at Week 52 with Omvoh were corticosteroid-free for at least 12 weeks (n=178/182).
12 weeks of continuous treatment includes the period of intermittent oral corticosteroid use during LUCENT-2.
- a Clinical remission was defined as an MMS subscore of stool frequency = 0 or 1 (with ≥1-point decrease from baseline), rectal bleeding = 0, and endoscopic = 0 or 1 (excluding friability).
- b Endoscopic improvement was defined as an MMS endoscopic subscore of 0 or 1 (excluding friability).
- c HEMR was defined as endoscopic remission plus histologic remission per Geboes scoring.
HEMR=histologic-endoscopic mucosal remission; MMS=modified Mayo Score; SC= subcutaneous; Q4W=every 4 weeks.
Among patients who achieved clinical response with Omvoh at Week 121
Omvoh demonstrated clinical remission regardless of biologic experience1
Bio-naïvea and Bio-failedb Patients Achieved Clinical Remission at Week 521
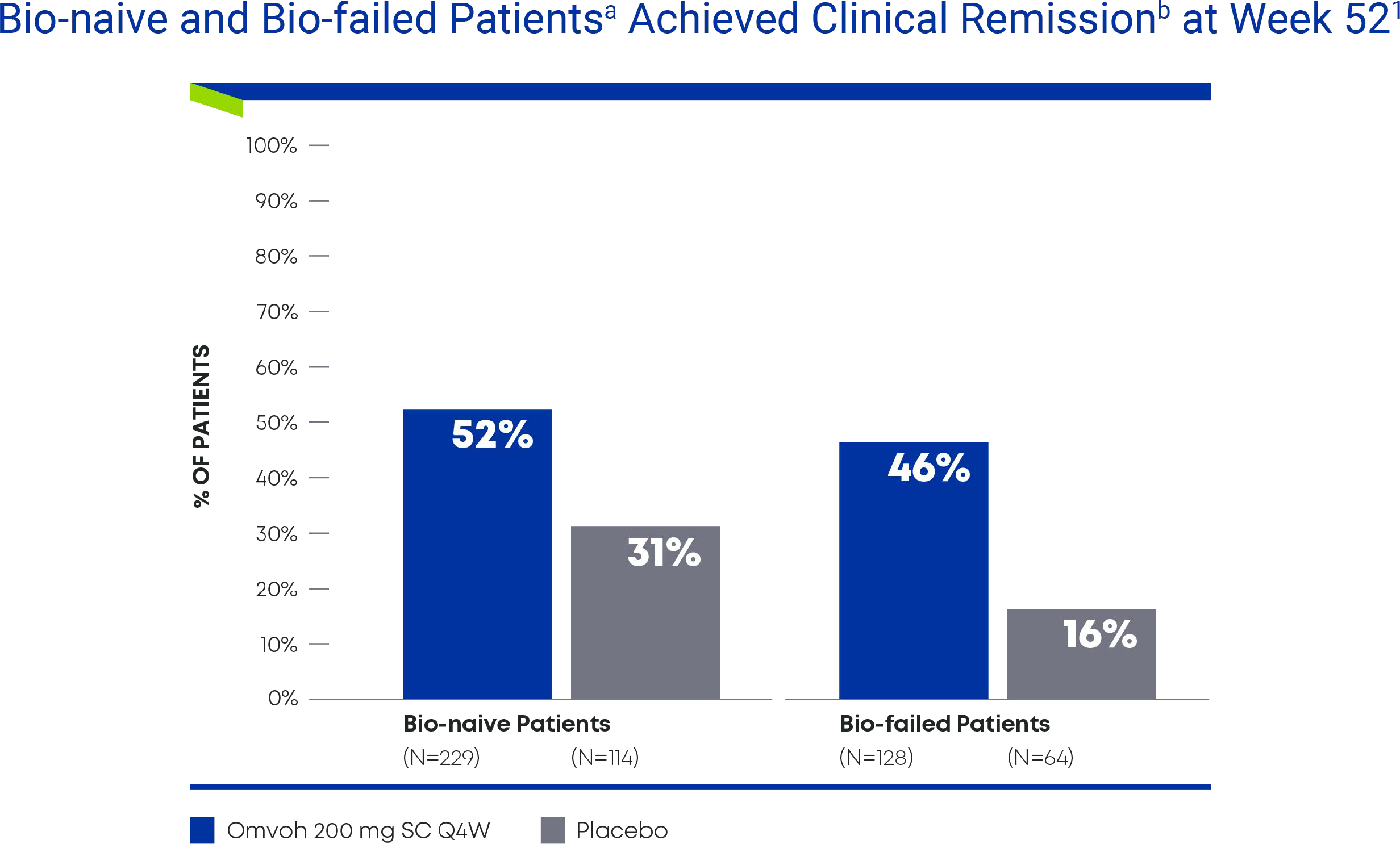
- a Bio-naïve includes patients who either had not been treated with biologics or were exposed to biologics but did not experience inadequate responses, loss of response, or intolerance.
- b Bio-failed includes prior biologic failures (inadequate response, intolerance to one or more biologic therapy).
- Prespecified subgroup analysis not controlled for multiplicity.
Bio-failed=biologic-failure; bio-naïve=biologic-naïve; SC= subcutaneous; Q4W=every 4 weeks.
For adults with moderately to severely active UC1
Omvoh achieved and sustained clinical remission up to 4 years1,2
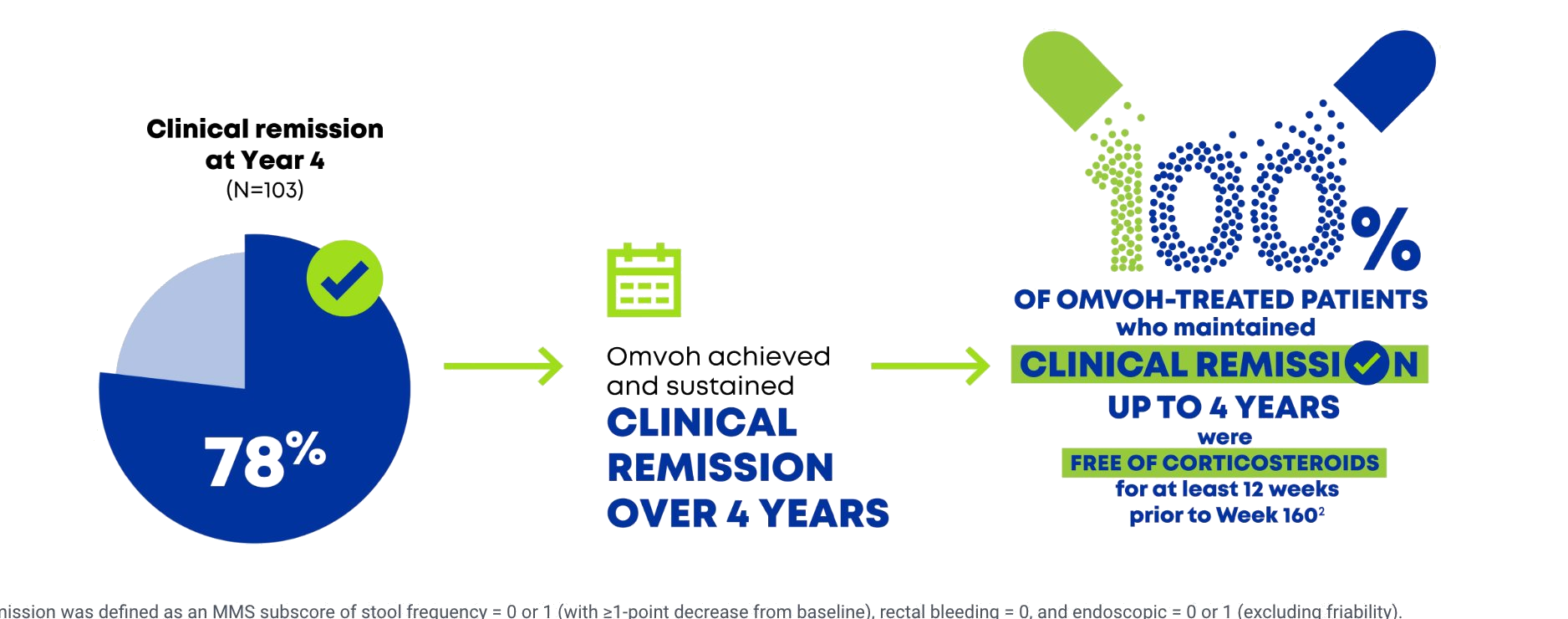
- Clinical remission was defined as an MMS subscore of stool frequency = 0 or 1 (with ≥1-point decrease from baseline), rectal bleeding = 0, and endoscopic = 0 or 1 (excluding friability).
- Observed case (OC) population: patients who completed Week 160 of continuous Omvoh treatment.
MMS=modified Mayo Score; OC= observed case.
For adults with moderately to severely active UC1
Patients achieved clinical, endoscopic and BU remission, along with HEMR up to 4 years1,2
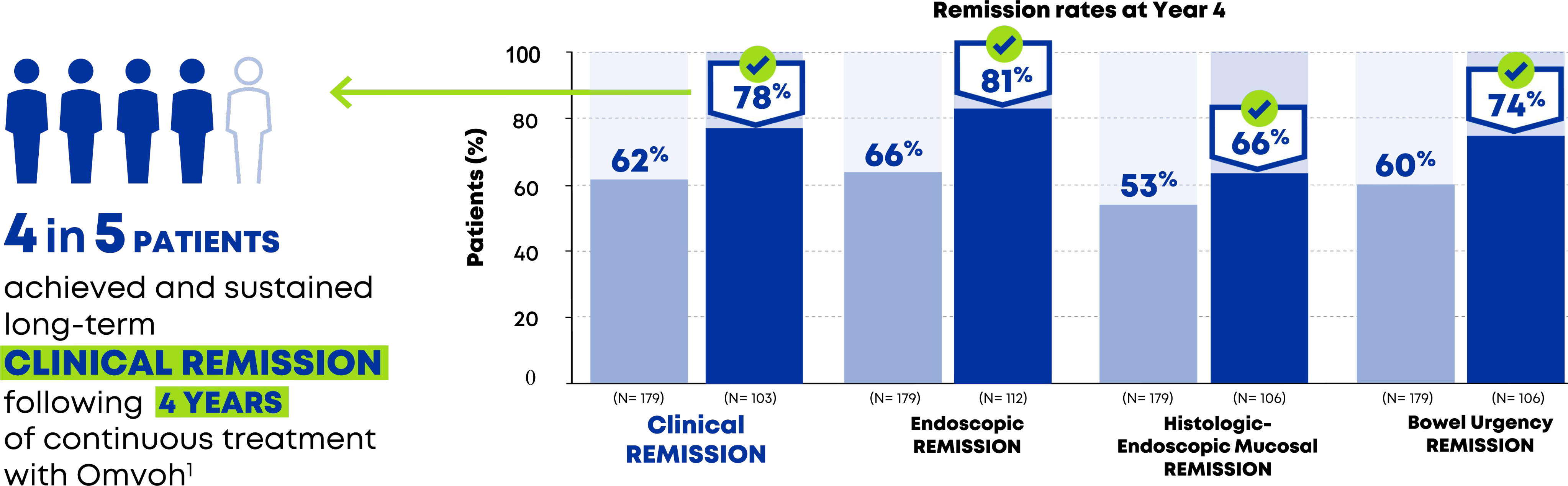
- Efficacy at Week 212 — endoscopic remission, histologic-endoscopic mucosal remission (HEMR), and bowel urgency remission shown for Week 12 remitters.
- Observed case (OC) analysis. Prespecified secondary and exploratory endpoints not controlled for multiplicity.
BU=bowel urgency; HEMR= histologic-endoscopic mucosal remission; OC=observed case.
For adults with moderately to severely active UC1
Omvoh significantly improved bowel urgency as early as Week 2 vs placebo, sustained up to 4 years1–3
Analysis was completed for patients in the mITT population to estimate the mean change from induction baseline.
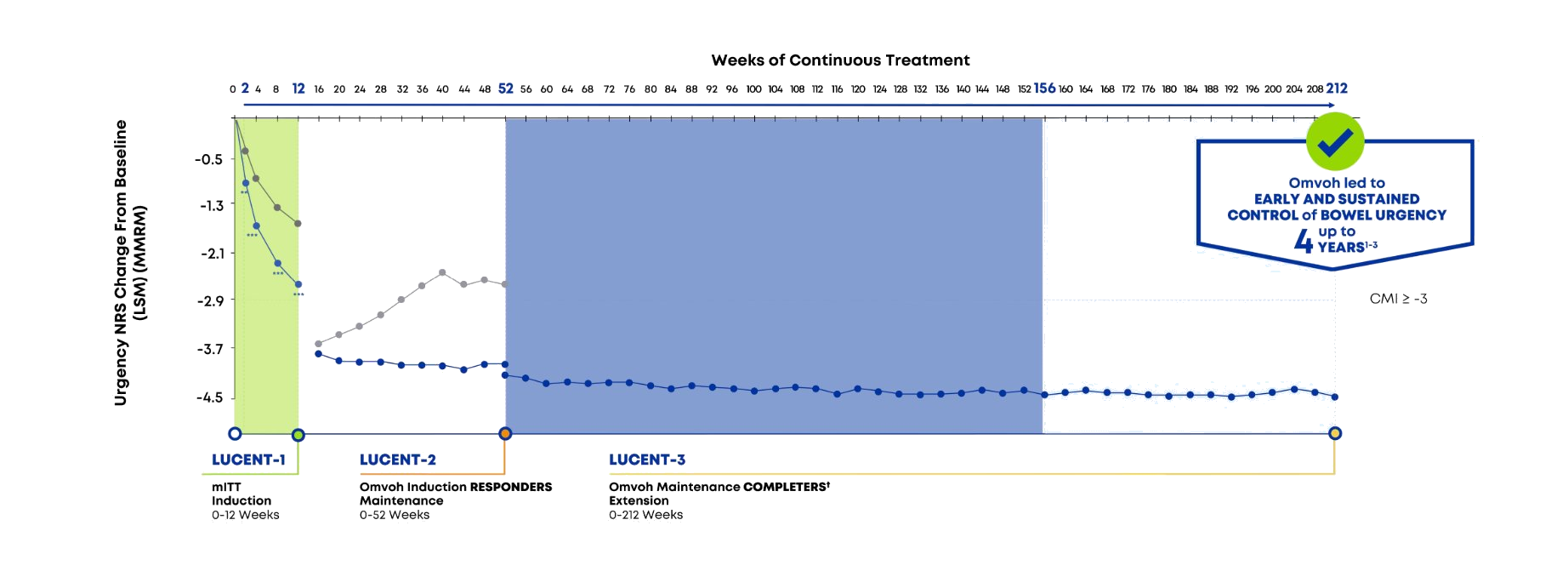
- MMRM was used for treatment comparison adjusting for baseline stratification factors. LSM were reported for each treatment group except for W0 of maintenance (W12 continuous treatment); no treatment comparisons in LUCENT-3.2
- Patients who were induction responders and who moved into LUCENT-3.2
CMI=clinically meaningful improvement; LSM=least squares mean; mITT= modified intent-to-treat; MMRM=mixed-effect model of repeated measures; NRS=numeric rating scale; PBO=placebo.
BU = bowel urgency; HEMR = histologic-endoscopic mucosal remission; IV = intravenous; LSM = least squares mean; mITT = modified intent-to-treat; MMRM = mixed-effect model of repeated measures; MMS = modified Mayo Score; NRS = numeric rating scale; OC = observed case; PBO = placebo; Q4W = every 4 weeks; RB = rectal bleeding; SC = subcutaneous; SF = stool frequency; UC = ulcerative colitis.
- Omvoh EUSPC FEB 2025.
- Sands BE, et al. Inflamm Bowel Dis. 2025;31(7):1876–1890.
- D'Haens G, Dubinsky M, Kobayashi T, et al. Mirikizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med.2023;388(26):2444–2455.
