Omvoh safety profile across the UC and CD patient populations
Omvoh has an established and consistent safety profile across UC and CD1–4
Most Common Treatment-Emergent Adverse Events in LUCENT-1, -2, and -31,2,a
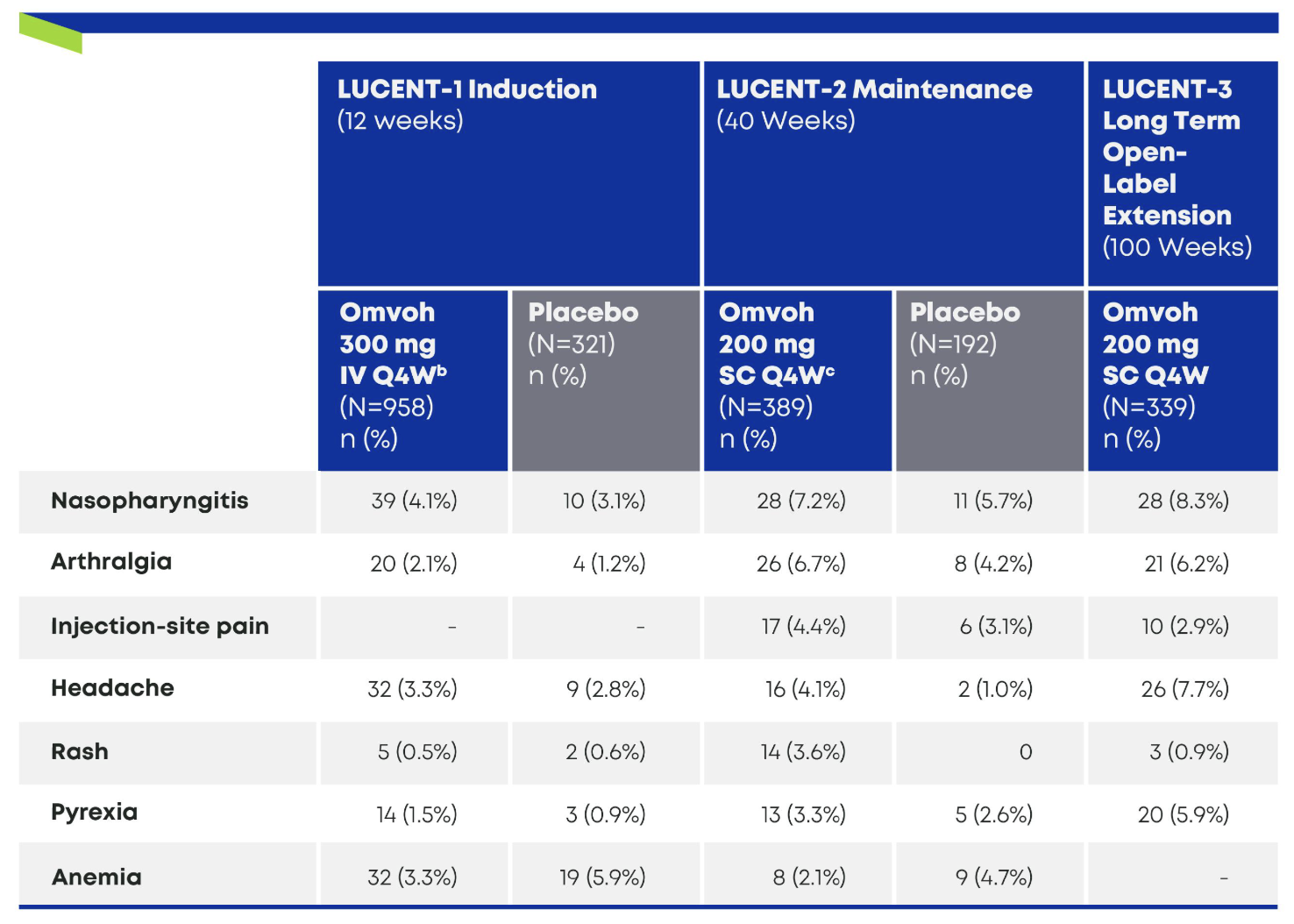
- a Common adverse events were defined as those that occurred in at least 3% of the patients in any trial group during the induction or maintenance trial. The events are listed according to decreasing frequency in the Omvoh group during the maintenance trial.1
- b Omvoh 300 mg as an intravenous infusion at Weeks 0, 4, and 8.1
- c Omvoh 200 mg as a subcutaneous injection at Week 12 and every 4 weeks thereafter for up to an additional 40 weeks.1
Most Common Treatment-Emergent Adverse Events in VIVID-1 and -23,4,a
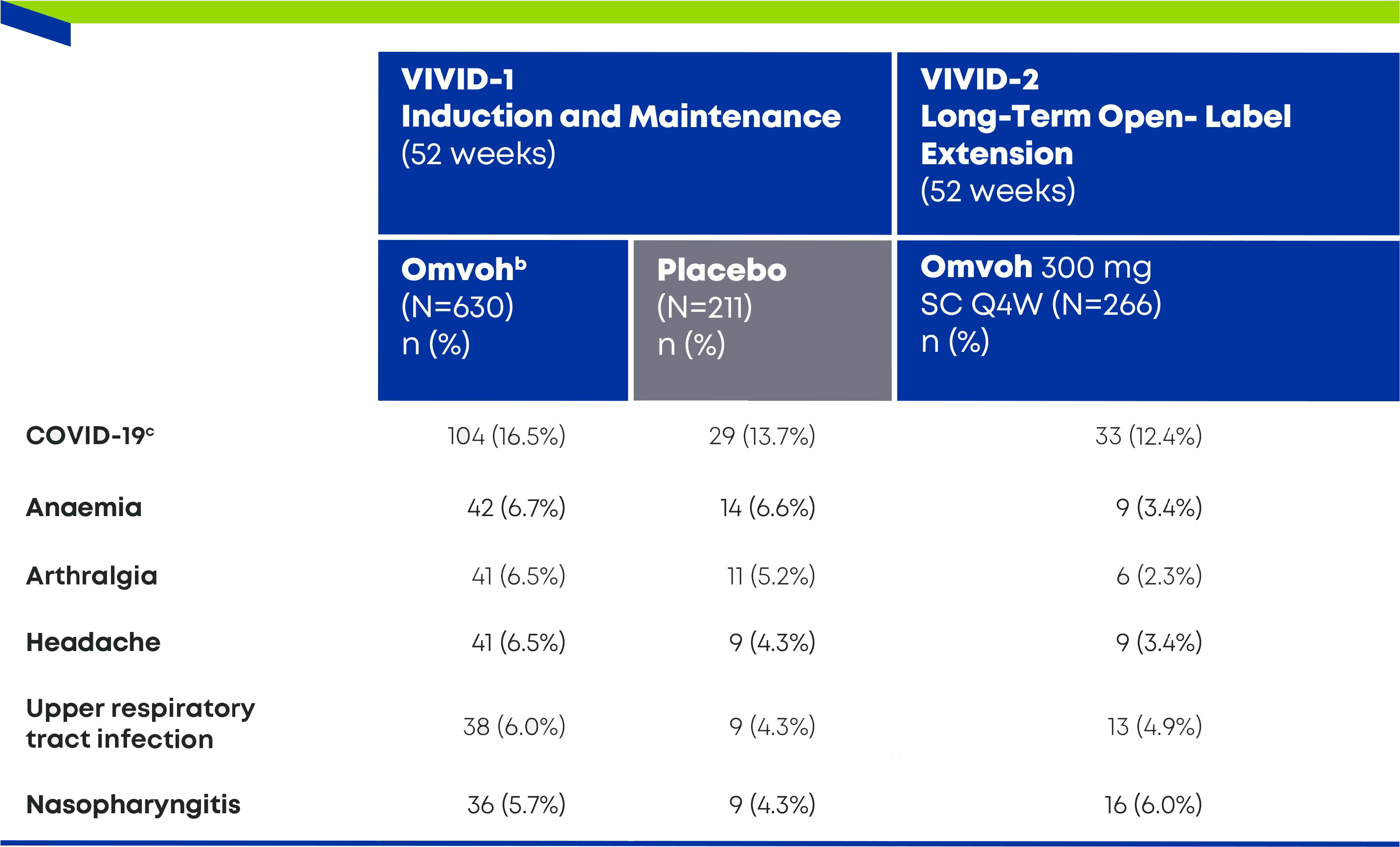
- a Common adverse events were defined as those that occurred in at least 3% of the patients in any trial group during the induction or maintenance trial. The events are listed according to decreasing frequency in the Omvoh group during the maintenance trial.3
- b Omvoh 300 mg as an intravenous infusion at Weeks 0, 4, and 8.3
- c Omvoh 200 mg as a subcutaneous injection at Week 12 and every 4 weeks thereafter for up to an additional 40 weeks.3
CD = Crohn's disease; UC = ulcerative colitis.
- D'Haens G, Dubinsky M, Kobayashi T, Irving PM, Howaldt S, Pokrotnieks J, et al. Mirikizumab as induction and maintenance therapy for ulcerative colitis. The New England Journal of Medicine. 2023 Jun 29; 388(26):2444–2455. Available from: https://doi.org/10.1056/nejmoa2207940.
- Sands BE, D'Haens G, Clemow DB, Irving PM, Johns JT, Gibble TH, et al. Three-year efficacy and safety of mirikizumab following 152 weeks of continuous treatment for ulcerative colitis: results from the LUCENT-3 open-label extension study. Inflammatory Bowel Diseases. Published online October 25, 2024; Epub: izae253 (Incl Suppl Mat). Available from: https://doi.org/10.1093/ibd/izae253.
- Ferrante M, D'Haens G, Jairath V, Danese S, Chen M, Ghosh S, et al. Efficacy and safety of mirikizumab in patients with moderately-to-severely active Crohn's disease: a phase 3, multicentre, randomised, double-blind, placebo-controlled and active-controlled, treat-through study. Lancet. 2024;404(10470):2423–2436. Available from: https://doi.org/10.1016/S0140-6736(24)01762-8.
- Barnes E, Sands B, D'Haens G, Hisamatsu T, Kelly C, Lu N, et al. Long-term efficacy and safety of mirikizumab following 104 weeks of continuous treatment for Crohn's disease: results from the VIVID-2 open-label extension study. Inflammatory Bowel Diseases. 2025;31(1):S10. Available from: https://doi.org/10.1093/ibd/izae282.024.
