Dosing
For adults with moderately to severely active UC1
Induction Phase
Maintenance Phase
- Initially intravenous infusion, then subcutaneous maintenance injections every 4 weeks, starting at Week 12.1
- Patients can administer Omvoh with ease and convenience through prefilled pens for maintenance dosing.1
Omvoh is intended for use under the guidance and supervision of a healthcare professional. Patients may self-inject after training in proper technique.1
aFor at least 30 minutes.1
bPreparation of 3 × 300 mg per IV infusion dose for at least 90 minutes.
CD=Crohn's disease; UC=ulcerative colitis.
1. Omvoh EUSPC FEB 2025.
Flexible Start With Omvoh In UC1,2

- † Omvoh responders in LUCENT-1=551/868 (63.5%); 544 entered LUCENT-2.1,2
- ‡ Patients with loss of therapeutic response during maintenance treatment may receive Omvoh 300 mg IV infusion every 4 weeks for a total of 3 doses (reinduction). If clinical benefit is achieved from this additional IV therapy, patients may resume Omvoh SC dosing every 4 weeks. The efficacy and safety of repeated reinduction therapy have not been evaluated.1
IV, intravenous; SC, subcutaneous; UC, ulcerative colitis.
1. Omvoh EUSPC FEB 2025. 2. D'Haens G, et al. Inflamm Bowel Dis. 2024;30(12):2335–2346.
Omvoh offers in-label flexible dosing to help to meet your patients' needs1,2
Extended Induction and Reinduction Dosing1
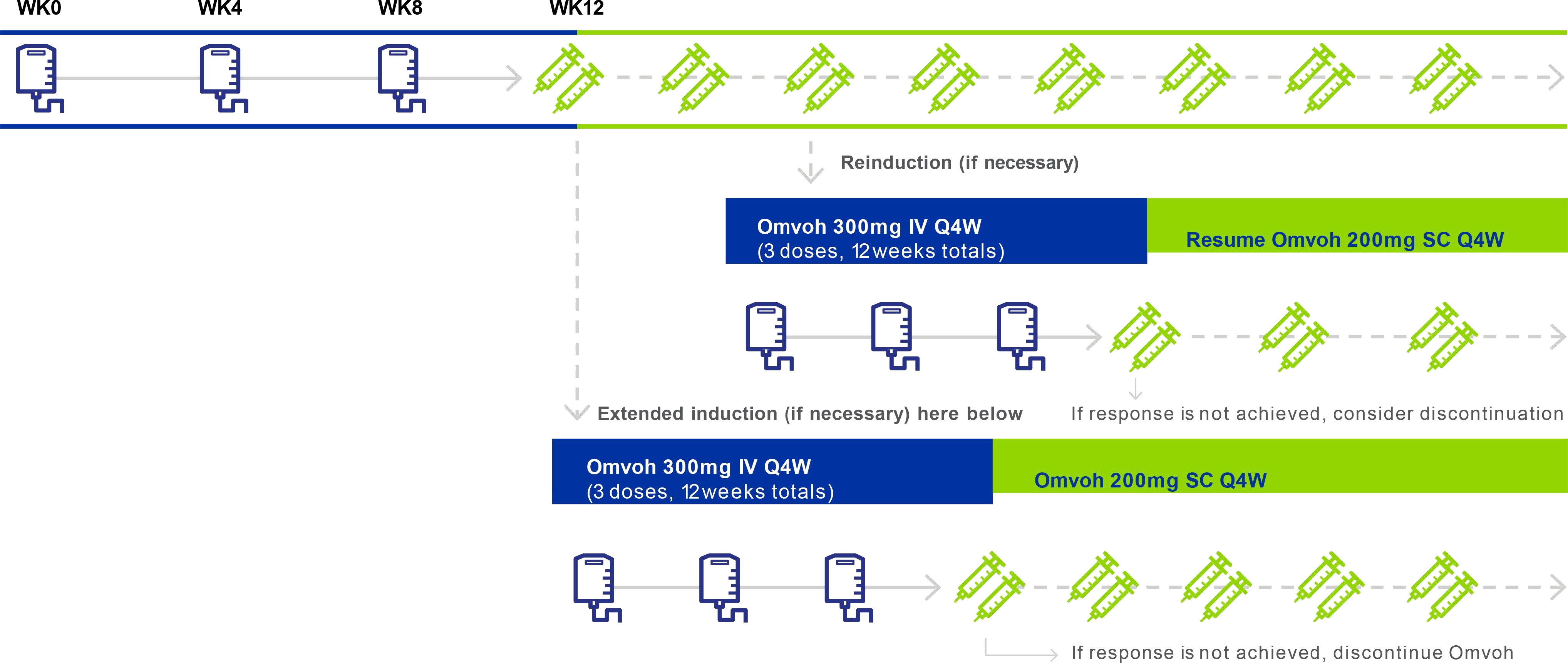
- Some patients may need extended induction therapy following initial induction to achieve clinical response.1
- If a patient loses response during maintenance therapy, reinduction therapy can help them regain response.1
IV, intravenous; Q4W, every 4 weeks; SC, subcutaneous.
1. Omvoh EUSPC FEB 2025. 2. D'Haens G, et al. Inflamm Bowel Dis. 2024;30(12):2335–2346.
Omvoh Extended Induction Helps Week 24 Response Rates Among Week 12 Non-responders
Majority of patients treated with Omvoh 300 mg IV achieved clinical response at Week 12 (induction) or Week 24 (extended induction)1
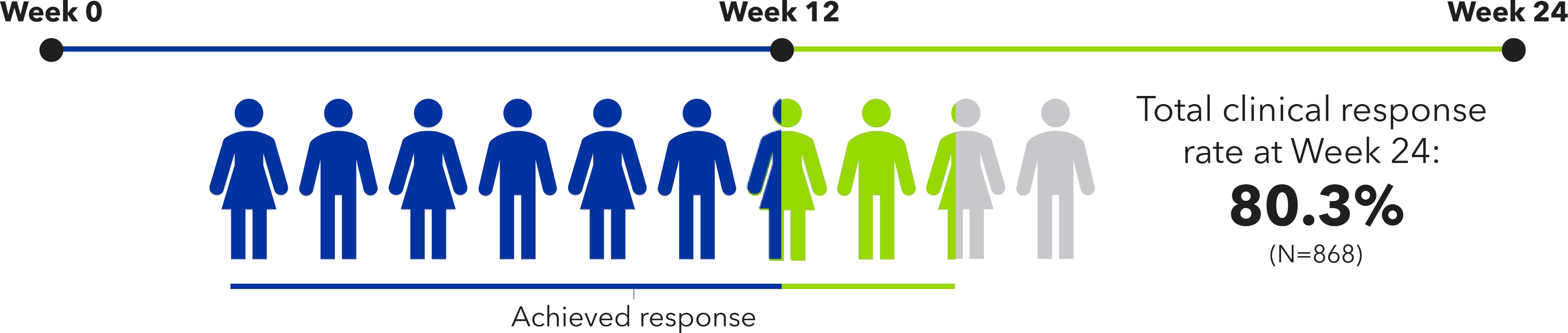
LUCENT-11
• 63.5% of patients achieved early clinical response with IV induction by Week 12 (N=868)
LUCENT-22
• 54% of patients who did not achieve adequate response at Week 12 went on to achieve clinical response at Week 24 with Omvoh extended induction therapy (N=272)
Clinical Response: ≥2-point and ≥30% decrease in the MMS from baseline; RB=0 or 1, or ≥1-point decrease from baseline.
IV, Intravenous; MMS, modified Mayo Score.
1. D'Haens G, et al. N Engl J Med.2023;388(26):2444–2455 (including supplementary index). 2. D'Haens G, et al. Inflamm Bowel Dis. 2024 Jan 25:izae004. Epub ahead of print.
In Total, 80% Of Omvoh-treated Patients Achieved Clinical Responsea At Week 24, Including Week 12 Non-responders Receiving Three Additional Doses1
Clinical response: NRI and mBOCF data1
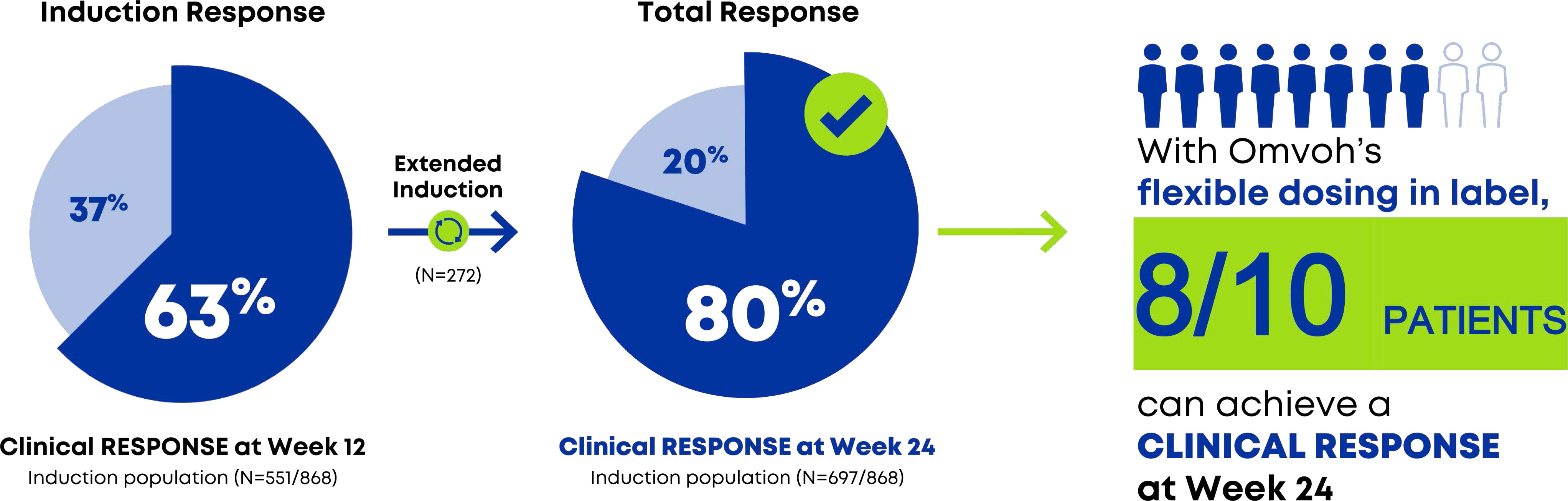
a Clinical remission was based on the MMS and was defined as: SF subscore=0 or 1 with a ≥1-point decrease from baseline, RB subscore=0, and ES=0 or 1 (excluding friability).1
Clinical response: ≥2-point and ≥30% decrease in the MMS from baseline: RB=0 or 1, or ≥1-point decrease from baseline.2
IV, intravenous; mBOCF, modified baseline observation carried forward; MMS, modified Mayo Score; NRI, non-responder imputation; RB, rectal bleeding; SC, subcutaneous.
1. D'Haens G, et al. Inflamm Bowel Dis. 2024;30(12):2335–2346. 2. D'Haens G, et al. N Engl J Med.2023;388(26):2444–2455 (+ Suppl. index).
